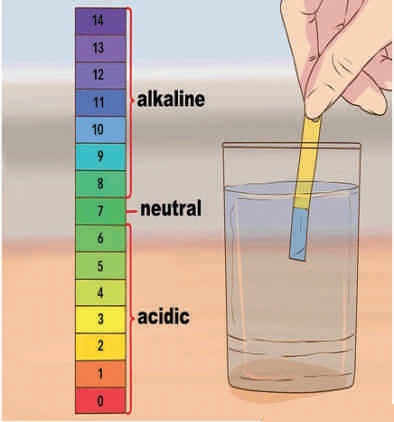
pH is generally understood in terms of acidity or basicity (to which people sometimes get confused with sour or salty) but it is a chemical characteristic which is affected by contents dissolved in the water and is determined by the concentration of hydrogen ions in it. pH is exhibited as a value ranging between zero to 14. Water pH is an important operational water quality parameter. It fluctuates with differing environmental factors including gases, chemicals and solids. The pH (acidity or basicity) of water effects its composition. The safe pH range for drinking water spans between 6.5 to 8.5. The WHO guidelines for drinking water quality do not propose health-based guideline value for it. The Central Pollution Control Board (CPCB) of India has prescribed Designated Best Use Water Quality Criteria according to which the pH for drinking water without conventional treatment but after disinfection has been prescribed between 6.5 and 8.5, whereas after conventional treatment and disinfection it can be between 6 to 9. For propagation of wildlife and fisheries the water pH may range between 6.5 to 8.5.
There are different methods for estimation of the pH like test by pH strip or titrimatric method or through electronic sensors etc.