Hardness of water is an important characteristic that is good if within permissible limits and not good if it is more than permissible. In our daily life we experience the hardness of water but mostly unable to relate the experience with the characteristics and its effect. There are simple means by which one can observe and understand as well as can test to know whether water from a source is ‘hard’ or not. We can determine the degree of hardness of any water sample by testing the foam/lather produced by it with soap and comparison with lather produced by known water sample which is not hard.
Soap or detergent powder, distilled water, mug or small bucket, 2 screw cap small vials (50 ml vials are sufficient for the test) / alternatively you can take any small bottles made of glass or transparent material, of same quantity which are washed well and dried).
1- Take soap and rub and mix it in distilled water. This will be used as soap water for producing lather.
2- Take a screw cap bottle and fill it up to ¼ volume with soap water and further fill another ¼ volume with distilled water. Lather produced in it will be used (as controlled) to compare with the lather produced in another bottle with tested water sample.
3- Take second bottle and fill it to ¼ volume with soap water and fill further same volume with sample water whose hardness is to be tested.
4- Shake both bottles equally for one minute.
5- Watch the amount of foam/lather produced in both bottles.
6- Compare the lather produced in second bottle (having sampled water) with that produced in first bottle (with distilled water).


Images: Comparison of lather production Images: Test of lather production in water
7- If the amount produced in both bottles is same, it would reflect that sampled water is soft.
8- If the foam produced is lesser then the corresponding water shall be considered having hardness.
9- If no foam/lather is produced at all then the corresponding water shall be considered as very hard (i.e. the amount of hardness would be more than 180 mg/liter).
10- Take more samples of water from different sources and compare the lather produced with the controlled sample and document the observations systematically.
Instead of soap you can take small quantity of detergent water for producing lather and follow the same procedure.
The observations of hardness test for different water samples may be documented comparatively in a tabular form. An example of the table format and sample entries is given here –
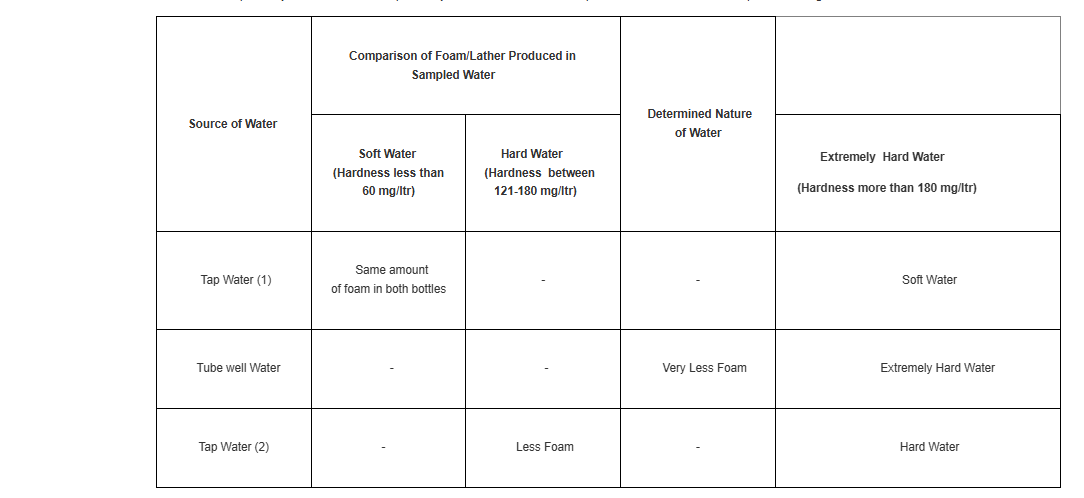
Write the comparative degree of hardness of different water samples through observations of foam produced. A database of hardness of water samples from different sources is prepared.
â— Concept of hardness of water.
â— Water from different sources may have different degree of hardness.
â— Water hardness can be understood by observation, and the degree of hardness can be ascertained by simple tests.
â— There are two broad categories of hardness in water – permanent and temporary, that are caused due to presence of different compounds. Permanent hardness is caused due to sulfates and phosphates of calcium and magnesium whereas temporary hardness is caused due to presence of their carbonates and bicarbonates.
â— How can hardness of water be removed? Can you remove the hardness of water and test again the amount of foam produced from it?
â— Can we prepare hard water by mixing some substances? Try with adding salt or soda in soft water in different amounts and test the change in hardness through observation of lather produced.
â— In the control bottle, instead of distilled water try with RO water / rain water / battery water / condensed water to produce foam.