pH is the measure of the acidic or basic nature of a solution. Its value ranges between zero to 14. To know the acidic or basic nature of a solution a litmus paper (which is a filter paper that is treated with natural-soluble dye obtained from lichens) is used whereas to ascertain the range or value of acidity or basicity standard pH paper strip (which is a paper treated with mixture of indicators) is used. The pH paper strip is much useful as it can be used to test more accurate value or range of the pH.
Basic Concept of pH-
1. pH of Neutral Solution - If the pH of a solution is 7, it will be a neutral solution. Such solution shall not react with litmus or any other liquid indicator.
2. pH of Acidic Solution- All acidic solutions have pH less than 7. Therefore, whenever the pH of a solution is less than 7 it will be acidic in nature and it will turn the blue litmus to red, and will cause liquid indicators, methyl orange turn to pink and phenolphthalein to colourless.
3. pH of alkaline solution- All alkaline solutions have a pH greater than 7. Therefore, whenever the pH of a solution is greater than 7 it will be alkaline in nature and will turn the red litmus to blue, and will cause methyl orange turn to yellow and phenolphthalein turn to pink.
â— pH paper strips,
â— A small clean container of glass or metal in which the water sample can be taken.
1- Take the water sample whose pH is to be determined in the vessel,
2- Take a strip from the pH paper strip booklet,
3- Dip the pH paper strip into the sample water and take out immediately and let it dry for few seconds for any change of colour,
4- Observe the change in colour of pH paper and match it with the colour scale printed on the pH paper strip booklet.
5- The pH of sampled water will be corresponding to the pH value printed on pH booklet with which the changed colours of the tested pH strip matches.
Image: Dip the pH strip in sample water Image- Change in Colour of pH Strip
Use reference range and write the determined pH value in the appropriate cell of pH range, in a table whose suggested format is given below
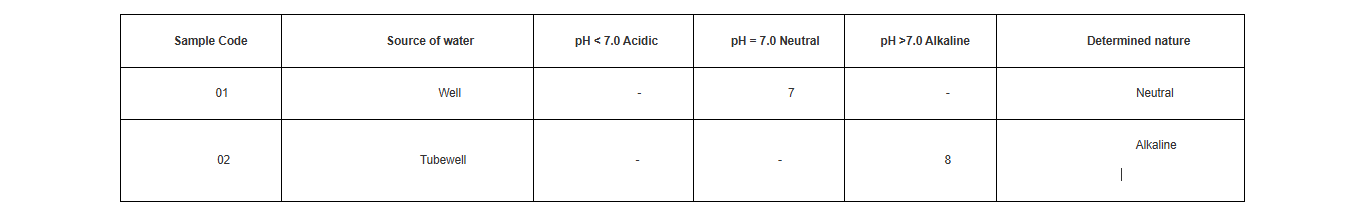
â— Systematically write the pH value observed for different samples of water.
â— Concept of pH related acidity, basicity and neutrality.
â— Classification of solutions based on acidic, basic and neutral nature.
â— You can test the acidic, basic or neutral nature of any solution by pH test.
â— pH of a solution is one of the factors that determine its reaction with other materials
â— Is it possible for you to make pH test paper strip? Explore how.
â— Why water samples from different sources show variations in acidity or basicity?
â— Mix the lime juice, soda and other substances in water and find out the pH value. Is any change is observed? If yes, why? By comparative analysis find out the reason of different pH observed in these solutions.
â— What is acid rain? How it occurs? Is there any alkaline rain too?